 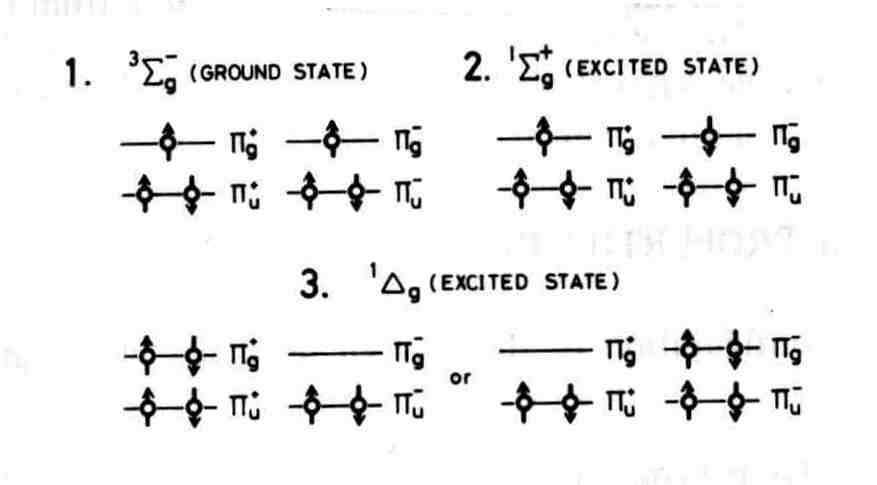 
Non-valence electrons are not represented in Lewis structures. The total number of electrons represented in a Lewis structure is equal to the sum of the numbers of valence electrons on each individual atom. Excess electrons that form lone pairs are represented as pairs of dots, and are placed next to the atoms.Īlthough main group elements of the second period and beyond usually react by gaining, losing, or sharing electrons until they have achieved a valence shell electron configuration with a full octet of (8) electrons, hydrogen (H) can only form bonds which share just two electrons.Ĭonstruction and electron counting Lines are drawn between atoms that are bonded to one another (pairs of dots can be used instead of lines). Lewis structures show each atom and its position in the structure of the molecule using its chemical symbol. 
Lewis structures extend the concept of the electron dot diagram by adding lines between atoms to represent shared pairs in a chemical bond. Lewis, who introduced it in his 1916 article The Atom and the Molecule. The Lewis structure was named after Gilbert N. A Lewis structure can be drawn for any covalently bonded molecule, as well as coordination compounds. Lewis structures, also known as Lewis dot formulas, Lewis dot structures, electron dot structures, or Lewis electron dot structures (LEDS), are diagrams that show the bonding between atoms of a molecule, as well as the lone pairs of electrons that may exist in the molecule. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
